Crystallising a new collaboration: Researchers reveal the structure of Leishmania donovani Rab5a protein
Congratulations to Network members Dr Ashish Arora (CSIR-CDRI, Lucknow, India) and Professor Ehmke Pohl (Durham University, UK) and their teams, on their first joint publication! Ashish and Ehmke met at the Network’s first HUB meeting in India, May 2018, and subsequently began collaborating to solve the crystal structure of the Leishmania donovani RAB5a protein, which is essential for parasite survival in its human host. Their work, funded via a NTD Network ‘Pump Priming’ allocation, establishes new links between their respective research institutions, and has supported the imitation of what Ashish and Ehmke intend as a long and fruitful partnership; one which could, in time, enable discovery of a new ‘druggable’ therapeutic target against visceral forms of leishmaniasis, a potentially lethal neglected tropical disease (NTD).
Visceral leishmaniasis: an urgent problem
Visceral leishmaniasis (VL) or kala-azar, is the most severe form of leishmaniasis, a neglected tropical disease (NTD) threatening the health of millions of people, particularly those living in poverty, in tropical areas worldwide. Two species of Leishmania parasites (the cause of infection) give rise to VL. The species commonly found in East Africa and across Asia is L. donovani, whilst in Europe, North Africa, and across Latin America, visceral disease results from L. infantum, also known as L. chagasi.1
VL infection is difficult to diagnose; the first clinical signs may be an enlarged liver and spleen, which are lethal if untreated. Current first-line treatments involve a month-long regimen of non-specific chemotherapeutics (pentavalent antimonials) which are themselves lethal for some patients. In endemic countries, people living in poverty typically cannot survive more than a few days without work, meaning that this therapy is ‘too expensive’ for these patients, even if available for free. There is an urgent need for new medicines that combat these parasites without harming patients. In order to develop such treatments, we need a much better understanding both of the parasite’s physiology, and of which Leishmania proteins could be usable as possible parasite-specific drug targets.

Leishmania parasites are spread to humans and other mammals (including livestock) by biting sandflies (Phlebotomus spp). This stained blood smear shows Leishmania infantum parasites (red arrow) in infected macrophages (white blood cells, white bar). The parasites damage host organs such as the liver and spleen, and tissues such as the bone marrow, which harbour large numbers of these immune system cells.
Image: Wikimedia Commons
Rab proteins: ‘shape-shifting’ enzymes allowing parasite survival
To develop a treatment targeted specifically to VL, we first need to identify a physiological process which is essential for their survival, and which uses bio-molecules found only in the parasites. The Leishmania parasites are at their most vulnerable during the transition from insect to mammal host, when they must swim free in the host’s blood stream, i.e. prior to invading a macrophage (white blood cell) and finding refuge in this host cell’s lysosomes (an inner cell compartment, or ‘organelle’, used to destroy invading viruses and bacteria). During the multiple stages of this transition, the parasites change both their physical shape and cellular biochemistry several times, both to counter the stresses imposed by their rapidly changing environment, and to obtain nutrients from their host’s blood.2
Leishmania Rab proteins are essential for surviving this transition. Rab ‘GTPases’ are enzymes found in all eukaryotic cells from yeast to mammals, with 30-75% homology (genetic similarity) across these taxa.3 They enable absorption (endocytosis) and movement of molecules inside cells (internal trafficking).
Rab proteins are ‘shape shifters’, shuttling between membrane-anchored active forms bound to GTP (guanosine triphosphate), and internal (cytosolic) inactive forms bound to GDP (guanosine diphosphate).4 Of these, Rab5 proteins are involved specifically with the earliest stages of endocytosis.5,6 In human cells, Rab5 endocytosis is linked with Caveolin-1,7 a cell scaffolding (cytoskeleton) protein, needed to form ‘caveolae’; tiny (50-80nm wide) cup-shaped pits in the cell membrane, and which form early in endocytosis. Intriguingly, this Caveolin-1 is also needed for the entry of L. donovani parasites into host macrophages.8

RAB proteins allow cells to absorb molecules from their environment by forming cup-shaped hollows in the cell’s outer membrane that then enclose and internalise the captured substances; a process known as endocytosis.
Image: Wikimedia Commons
L. donovani has two unique forms of Rab5b:9
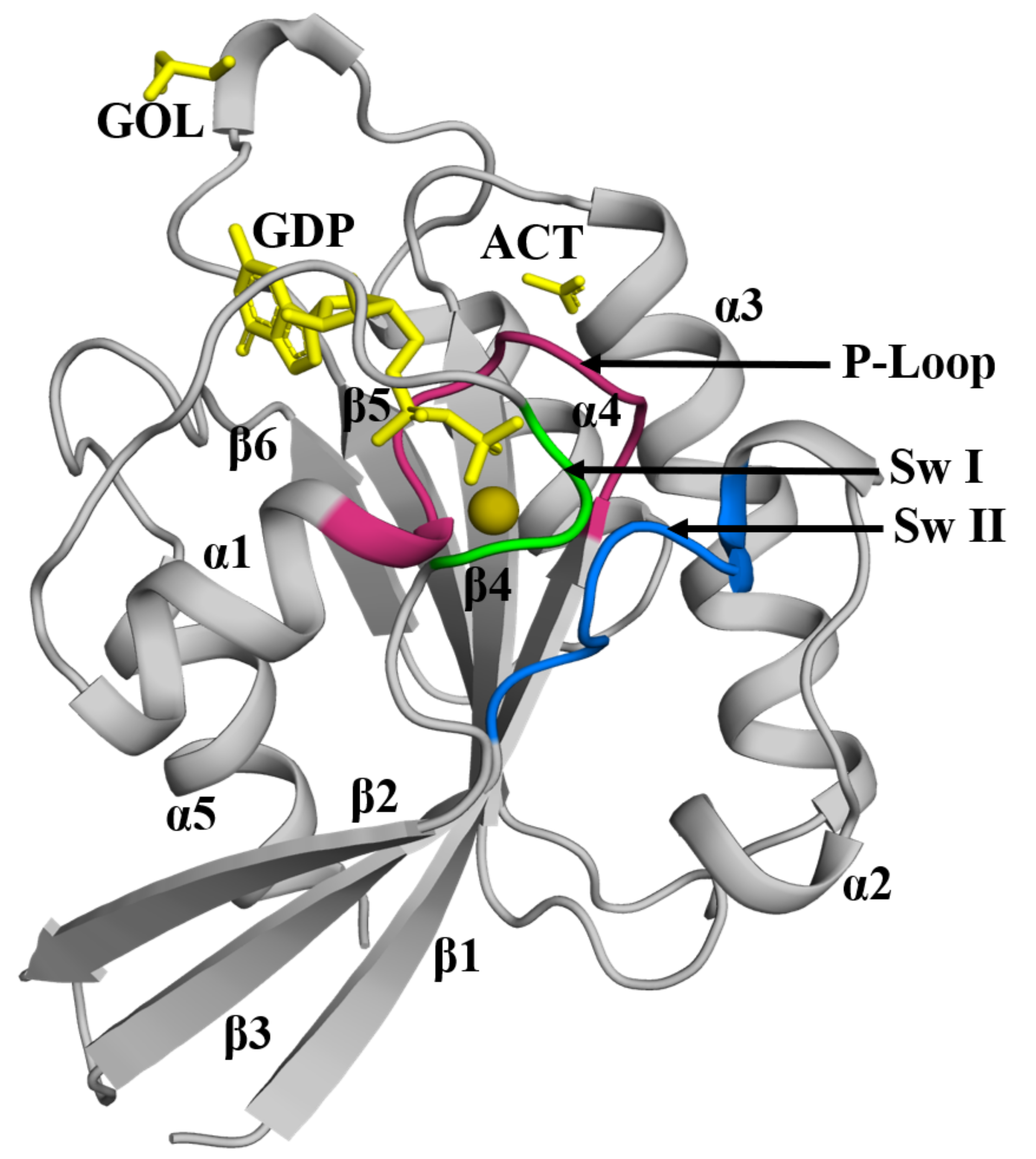
(a) Rab5a enables uptake of horseradish peroxidase and other external components, with roles in regulation of cell growth and targeting of the parasite to host cell lysosomes. (The image at the top of this article, taken from Ashish and Ehmke’s paper, shows the crystal structure of Rab5a from L. donovani, bound to GDP)
(b) Rab5b allows absorption of haemoglobin
Although the full physiological roles of these proteins is unclear, both are vital for the parasite to survive in its human host.9
The involvement of Rab5a in allowing parasites to enter the lysosomes of its host cells highlights this protein as a possible parasite-specific, druggable therapeutic target. However, to develop a drug which specifically affects Leishmania Rab5a, we need to identify which parts of the parasite protein deliver its vital functions in the parasite, and also which distinguish it from human forms of Rab5a. This requires detailed structural studies to compliment the physiological investigations.
Experts from Lucknow and Durham collaborate to reveal the secrets of Leishmania Rab5a
Ashish and Ehmke have brought together their respective expertises to determine the structure and function of Leishmania Rab5a protein by X-ray crystallography. The teams in Durham and Lucknow used molecular biology techniques to create forms of parasite protein with single amino acid changes, creating inactive mutant forms. Following crystallisation with guanosine nucleotides, these protein co-crystals were sent from India via Durham to the Diamond Light Source, in Oxfordshire where high-resolution data were collected. These experiment allowed the team to compare the structure of mutated protein forms with unmodified (active, wild-type) Rab5a. This has highlighted the domains implicated in the change of shape and electrostatic properties which shuttle the parasite protein between its active and inactive forms.
The unfolding story of a long-term collaboration
This analysis reveals that Leishmania Rab5a protein undergoes metamorphosis between active and inactive forms, through four distinct stages of folding;10 a metaphorical echo of the multiple stages of shape change which the parasite cells pass through as they transition from their insect to mammalian host. This data also reveals critical differences between Leishmania and human forms of Rab5a protein. This information is vital in order to identify portions of the Leishmania Rab5a protein that may be suitable for use as drug targets.
Although it is a great start, Ehmke and Ashish are aware that this work is only the beginning of their structure-based studies on Rab5a. We are still some way from verifying a target suitable for screening chemical compounds for a parasite-specific Rab5a inhibitor drug; the next stage in developing a novel treatment for VL. But as these determined researchers formulate their future plans, we look forward to further great insights arising from this inter-continental, collaborative friendship.
Read their publication here:
-
Muhammad Zohib, Diva Maheshwari, Ravi Kant Pal, Stefanie Freitag-Pohl, Bichitra Kumar Biswal, Ehmke Pohl and Ashish Arora. “Crystal structure of the GDP-bound GTPase domain of Rab5a from Leishmania donovani”. Acta Cryst 2020_F76_544-556. DOI: 1107/S2053230X20013722.

Professor Ehmke Pohl (back, second from left) and Dr Ashish Aurora (back, fourth from left) with NTD Network colleagues from India, South America and the UK, at the Taj hotel in Lucknow, during the Network’s first meeting in India, May 2018.
Image: Ehmke Pohl.
Mags Leighton
References
- Chappuis F, et al. Nature 2007; 5: 873–882.
- Marchese L, et al. Pathogens 2018; 7(2): 36.
- Zahraoui A., et al. J Biol Chem 1989; 264: 12394–12401.
- Vetter IR & Wittinghofer A. Science 2001; 294; 1299–1304.
- Bucci, C, et al. Cell 1992; 70, 715–728.
- Mukhopadhyay A, et al. J Cell Bio.1997; 136: 1227–1237.
- Hagiwara M, et al./Biochem Biophys Res Comm 2009; 378: 73–78.
- Kumar GA, et al. Nature Scientific reports 2019; 9(1): 1–11.
- Rastogi R, et al. J Boil Chem 2016; 291(28): 14737–14746.
- Zohib M, et al. Acta Cryst 2020; F76: 544–556.
